Recording date:
Duration:
Session:
Speaker:
Co-Authors:
Abstract:
Keywords
Hybrid, HMOs, reaction equilibrium, transmembrane transporters
Abstract
Major inherent limitations of enzyme catalyzed (in vitro) and whole-cell catalyzed (in vivo) biotransformation processes are the inability to change the reaction equilibrium position and ineffective product transportation out of cells, respectively. While in-situ product removal techniques can be used to shift a reaction equilibrium of an in vitro process it is predicated on substantial differences in chemo-physical properties between the substrate(s) and product(s), which is often not the case. Product export out of cells in an in vivo process can be achieved using transmembrane transporters. However, it is not trivial to find suitable transporters capable of exporting big and complex products. Moreover, they need to be very specific and not also export precursor molecules, which typically contain the same molecular structures recognized by the transporter binding sites. A novel technology that solves these inherent
limitations was developed by integrating in vitro and in vivo approaches into a hybrid process. The hybrid technology was proven to be efficient for the biomanufacturing of several different complex human milk oligosaccharides (HMOs) such as lacto-N-difucohexaose I (LNDFH-I), lacto-N-fucopentaose III (LNFP-III), lacto-N-fucopentaose II (LNFP-II), 3’-sialyl-3-fucosyllactose (FSL), sialyllacto-N-neotetraose (LST-c) and sialyllacto-N-tetraose (LST-a). Results from the hybrid production of a neutral (LNDFH-I) and a charged (LST-a) HMO are presented in this abstract. Figure 1 shows a schematic illustration of the hybrid process for the production of LNDFH-I.
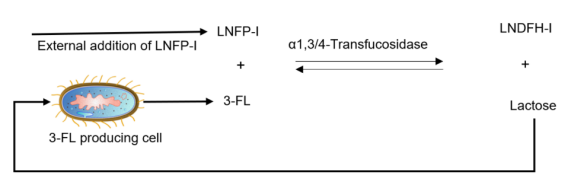
Figure 1: A schematic overview of hybrid process technology illustrating the production of LNDFH-I in a one-pot reaction by combining in vivo formation of 3-fucosyllactose (3-FL) and in vitro transfucosylation of lacto-N-ucopentaose I (LNFP-I) with in-situ recycling of the side-product lactose.
An in vitro synthesis of LNDFH-I by α1,3/4-transfucosidase catalyzed transfucosylation of lacto-N-fucopentaose I (LNFP-I) utilizing 3-fucosyllactose (3-FL) as a fucosyl donor led to 43% conversion of the supplied LNFP-I and producing a mixture at equilibrium consisting of 15% 3-FL,17% lactose, 35% LNFP-I and 33% LNDFH-I (wt./wt.). In comparison a hybrid process, which combined the in vivo 3-FL formation from lactose by fermentation and ex vivo transfucosylation of externally added LNFP-I into LNDFH-I by the action of the same α-1,3/4-transfucosidase, achieved 99% conversion of the supplied LNFP-I and produced a final mixture consisting of 37% 3-FL, 0.7% lactose, 0.8% LNFP-I and 61% LNDFH-I (wt./wt.). The high conversion in the hybrid process could be achieved as the enzymatic reaction equilibrium was circumvented by in-situ recycling of the side-product lactose (released in the
enzymatic step) into the fucosyl donor 3-FL. An in vitro LST-a process catalyzed by a α-2,3-transsialidase using lacto-N-tetraose (LNT) as acceptor and 3’-sialyllactose (3’-SL) as sialyl donor achieved 57% conversion of the supplied 3’-SL and produced a mixture at equilibrium consisting of 18% lactose, 21% 3’-SL, 18% LNT and 43% LST-a (wt./wt.). A corresponding hybrid process using the same α-2,3-transsialidase which combined the in vivo
production of LNT from lactose with the enzymatic transsialylation of the formed LNT using externally supplied 3’SL as sialyl donor, achieved full conversion of the 3’-SL and produced a final mixture of 35% LNT and 65% LST-a (wt./wt.), with no residual lactose or 3’-SL.